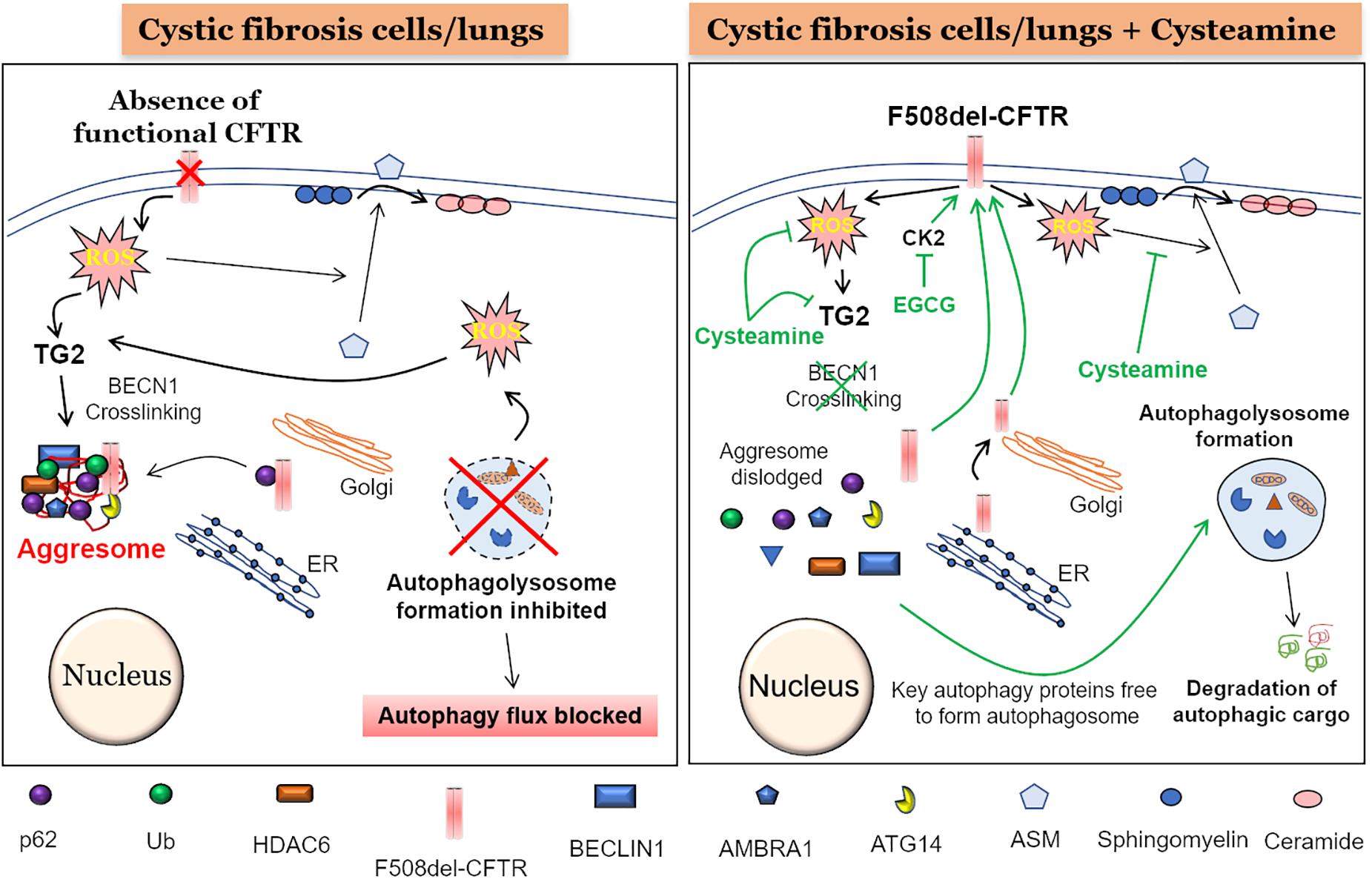
Frontiers | Adapting Proteostasis and Autophagy for Controlling the Pathogenesis of Cystic Fibrosis Lung Disease | Pharmacology
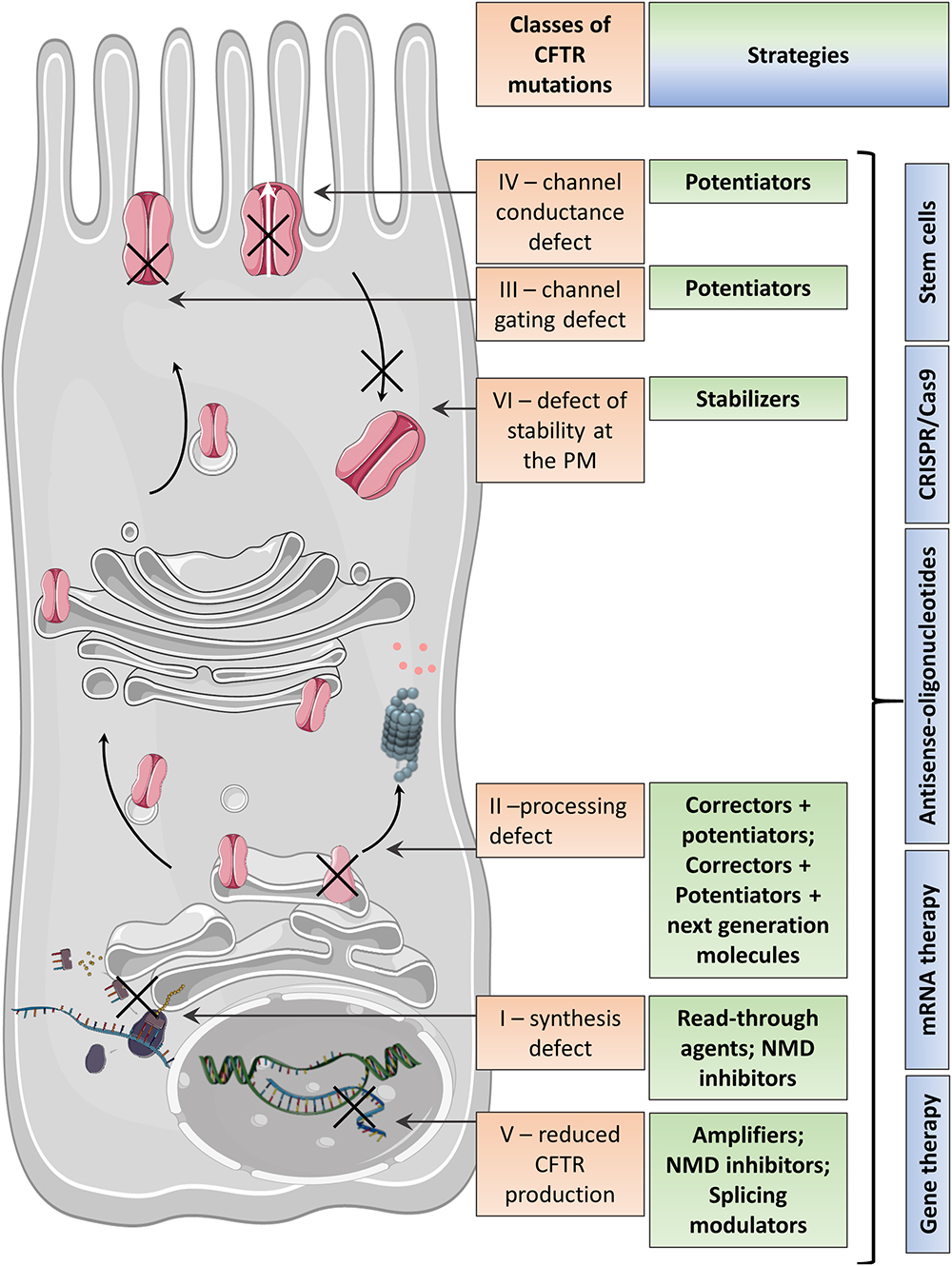
Frontiers | Emerging Therapeutic Approaches for Cystic Fibrosis. From Gene Editing to Personalized Medicine | Pharmacology

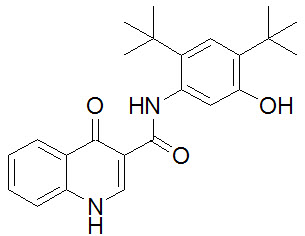
CFTR: New insights into structure and function and implications for modulation by small molecules - Journal of Cystic Fibrosis
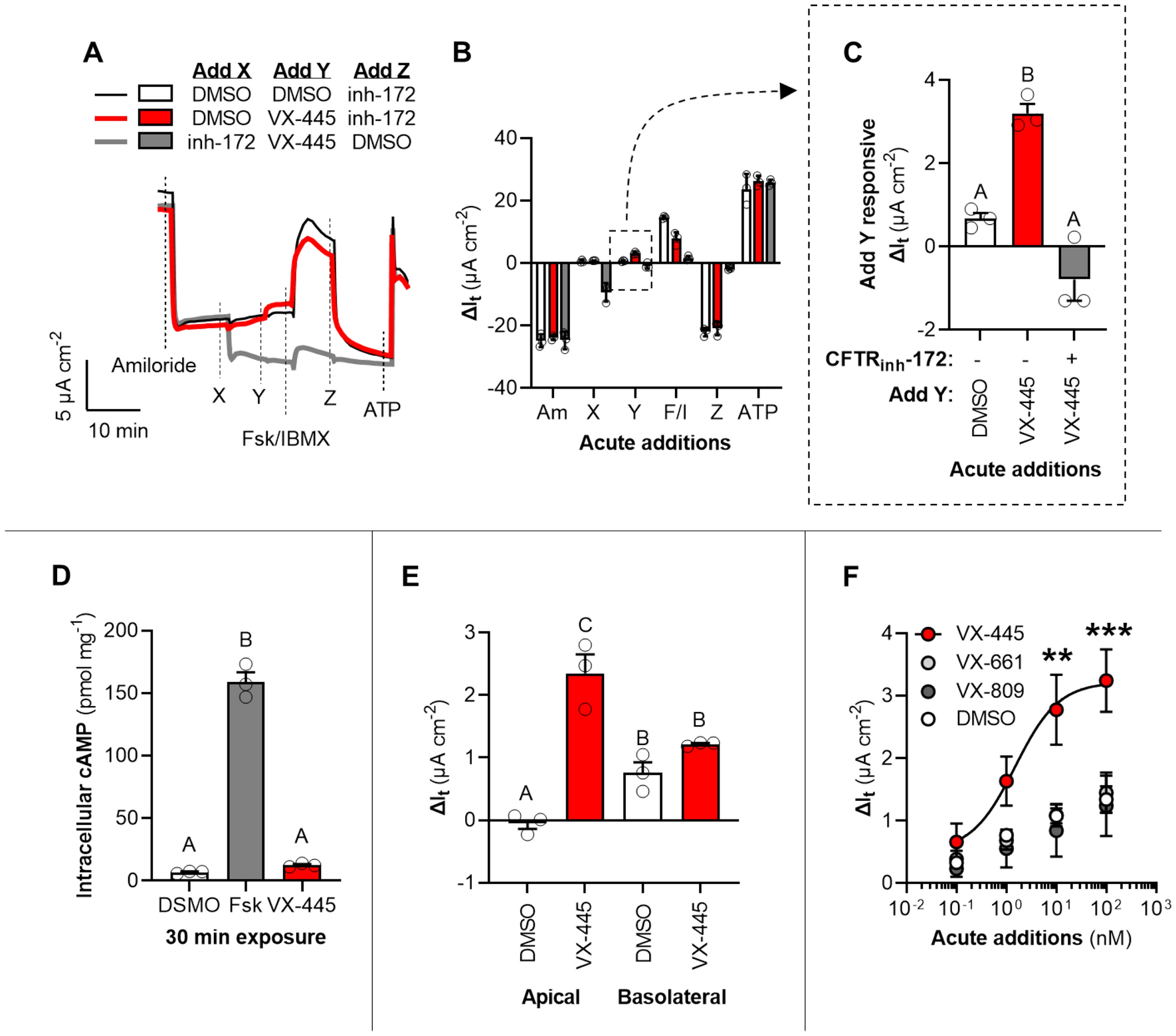
Elexacaftor is a CFTR potentiator and acts synergistically with ivacaftor during acute and chronic treatment | Scientific Reports
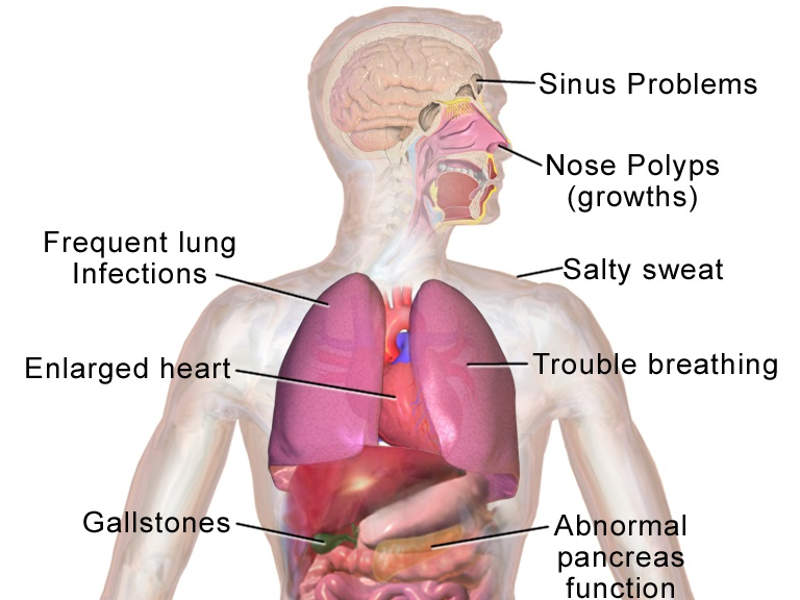
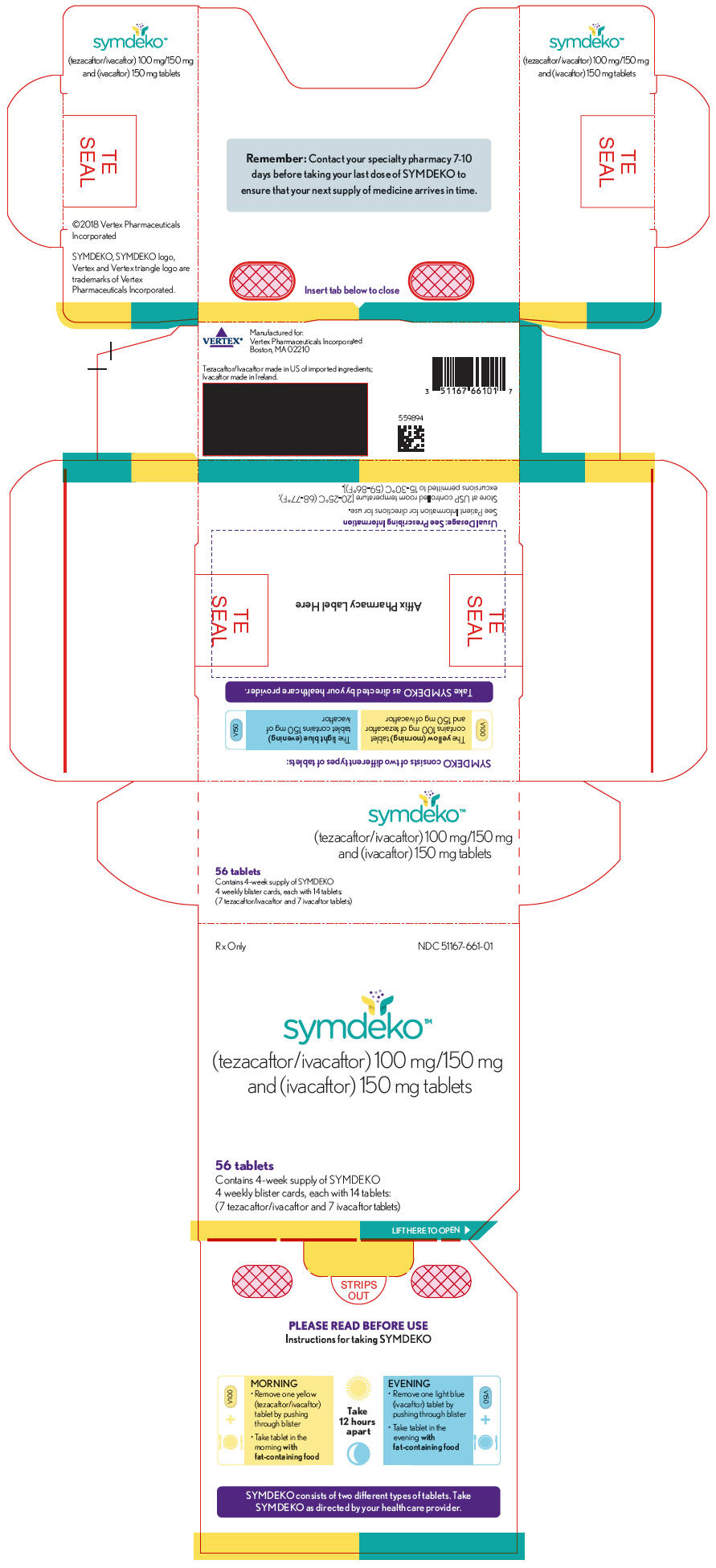
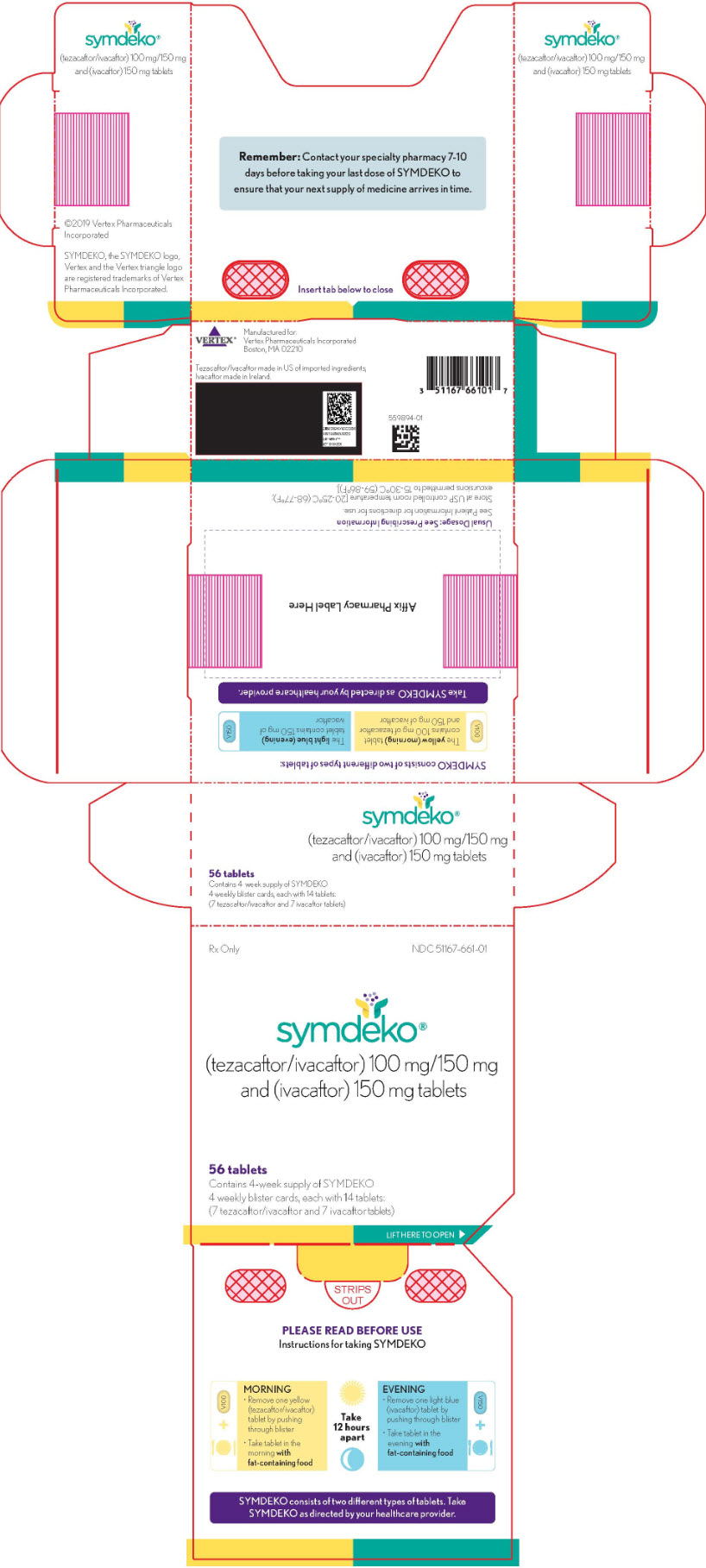
Symdeko (tezacaftor/ivacaftor and ivacaftor) for the Treatment of Cystic Fibrosis - Clinical Trials Arena

Impact of CFTR modulator use on outcomes in people with severe cystic fibrosis lung disease | European Respiratory Society
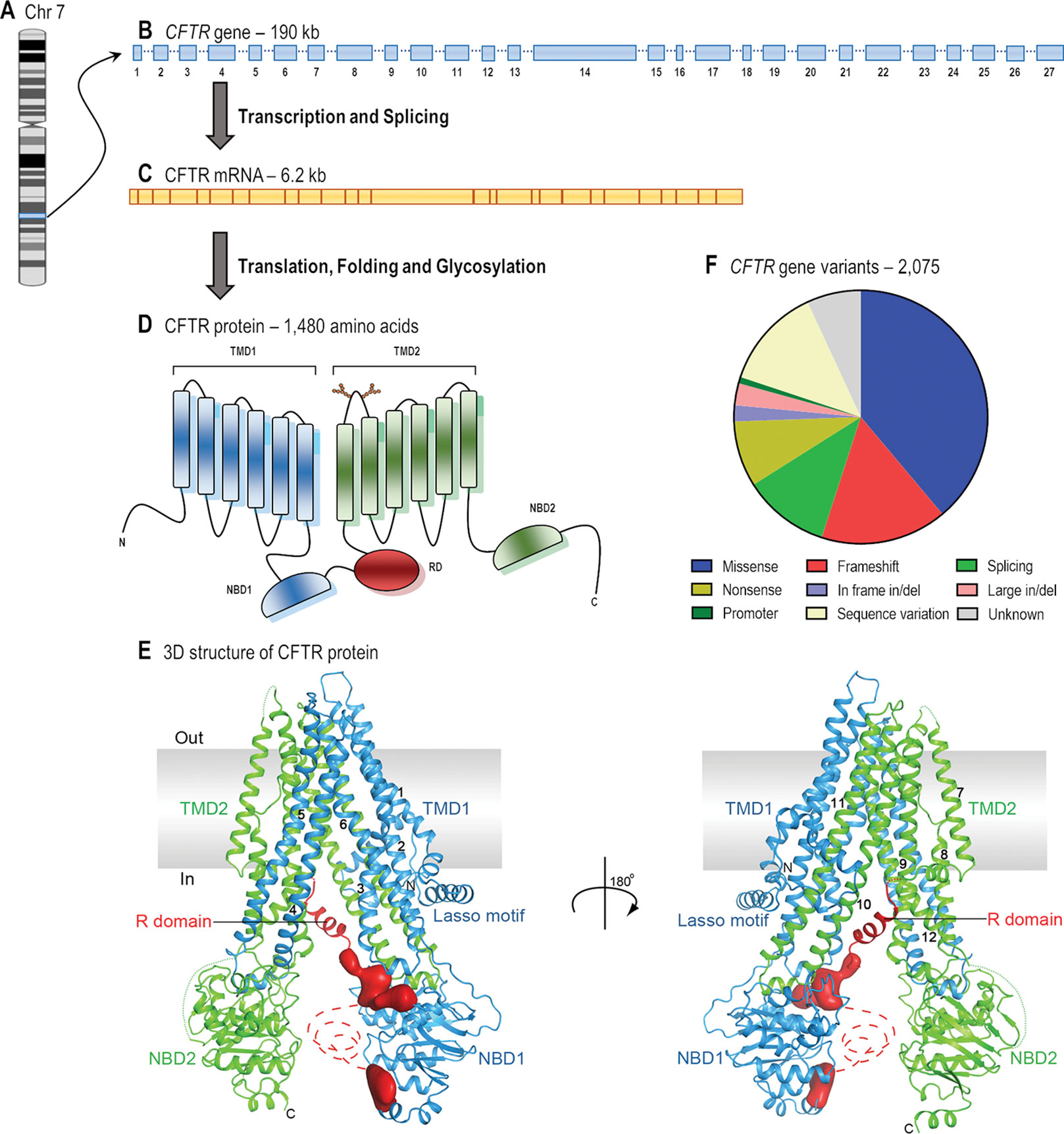
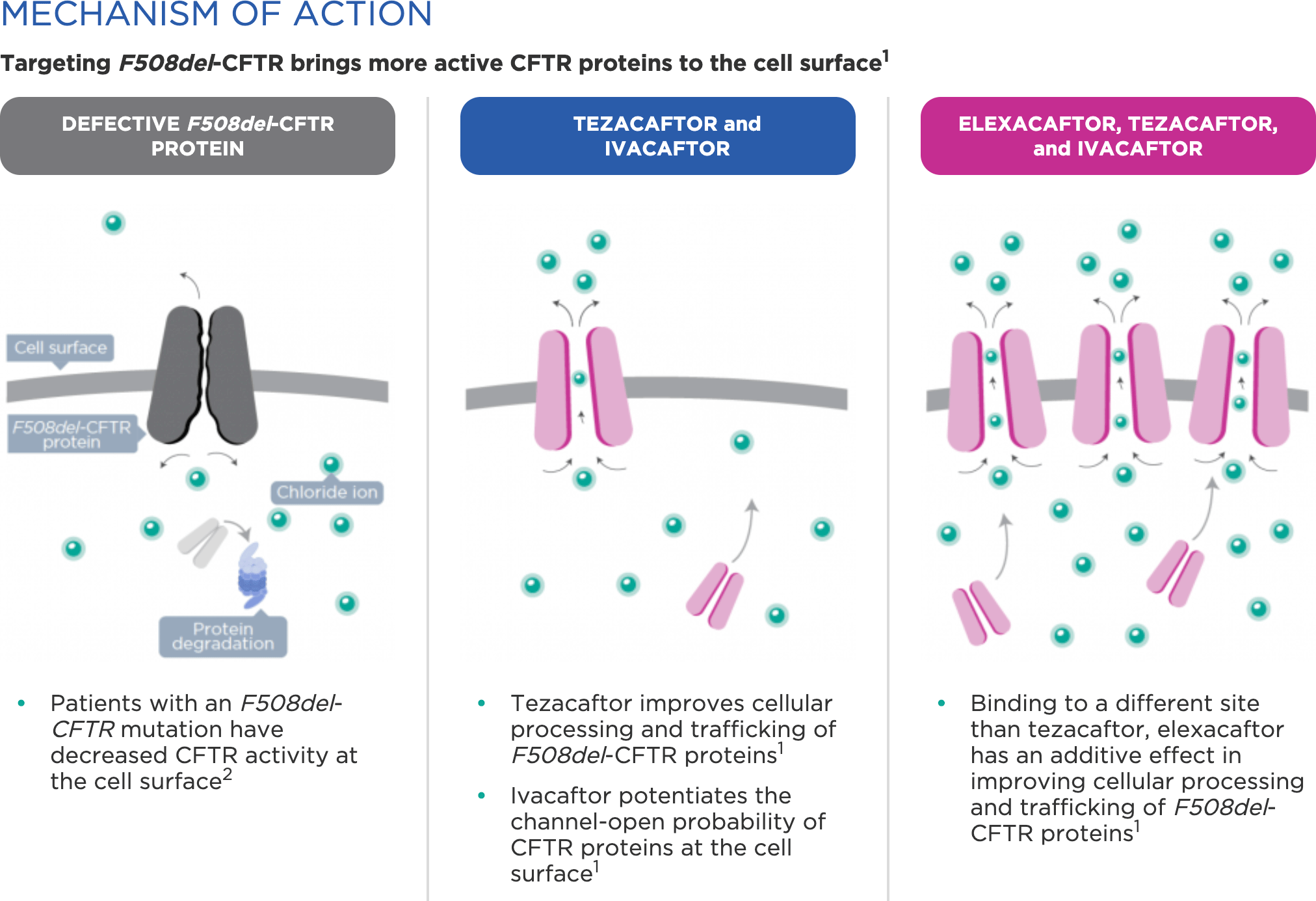
Frontiers | CFTR Modulators: The Changing Face of Cystic Fibrosis in the Era of Precision Medicine | Pharmacology
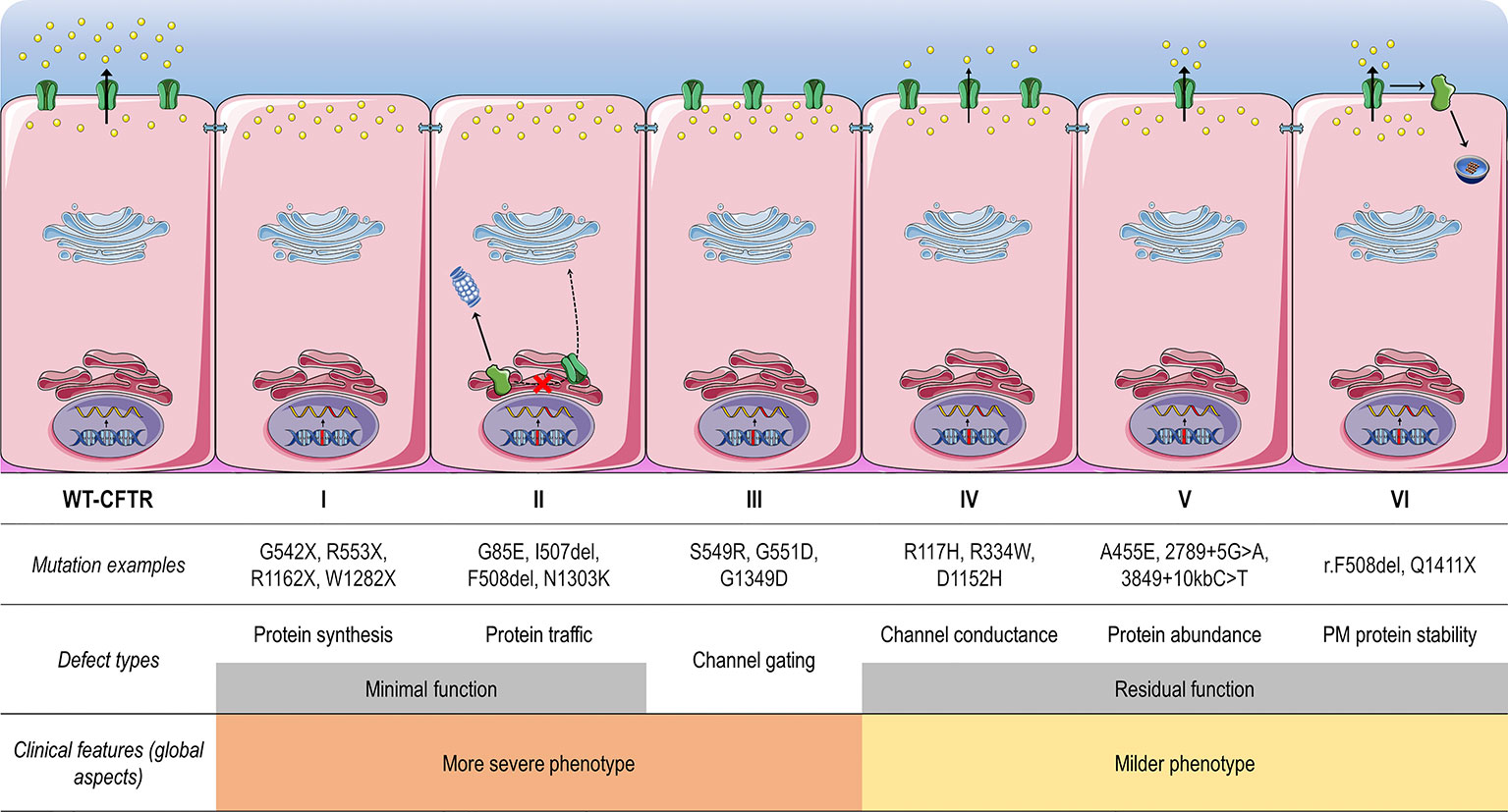
Frontiers | CFTR Modulators: The Changing Face of Cystic Fibrosis in the Era of Precision Medicine | Pharmacology

Elexacaftor-Tezacaftor-Ivacaftor: The First Triple-Combination Cystic Fibrosis Transmembrane Conductance Regulator Modulating Therapy. - Abstract - Europe PMC

FDA Approves SYMDEKOTM (tezacaftor/ivacaftor and ivacaftor) to Treat the Underlying Cause of Cystic Fibrosis in People Ages 12 and Older with Certain Mutations in the CFTR Gene | Business Wire

FDA Approves SYMDEKOTM (tezacaftor/ivacaftor and ivacaftor) to Treat the Underlying Cause of Cystic Fibrosis in People Ages 12 and Older with Certain Mutations in the CFTR Gene | Business Wire